Industry Standards for Peptide Suppliers: Ensuring Research Integrity
- Colt Clemons

- Feb 18
- 7 min read

Finding trustworthy peptide suppliers often becomes a crucial stage for American researchers pursuing high-impact experiments. Consistent results depend on the ability to source peptides that meet strict quality benchmarks, including purity, identity, and compliance with regulatory frameworks outlined by organizations like the United States Pharmacopeia. This article clarifies how industry standards shape supplier reliability, helping you navigate critical dimensions like synthesis methodology, impurity management, and the importance of rigorous quality management systems for reproducible research outcomes.
Table of Contents
Key Takeaways
Point | Details |
Industry Standards Are Crucial | Adhering to rigorous quality standards in peptide synthesis ensures product integrity and research reliability. |
Thorough Supplier Evaluation Is Essential | Researchers must meticulously assess suppliers based on documentation, testing protocols, and quality management systems. |
GMP and ISO Compliance Are Mandatory | Compliance with Good Manufacturing Practice and ISO standards is vital to maintain product quality and scientific integrity. |
Documentation Ensures Credibility | Comprehensive record-keeping and traceability are fundamental for reproducibility and accountability in research. |
Defining Industry Standards for Peptide Suppliers
Research peptide suppliers operate within a complex regulatory landscape that demands rigorous quality control and documentation. Comprehensive manufacturing guidelines have emerged to establish critical benchmarks for ensuring product integrity and research reliability.
Industry standards for peptide suppliers typically encompass several key dimensions:
Synthesis Methodology: Defining precise chemical processes and reaction conditions
Purity Assessment: Establishing minimum acceptable purity thresholds (typically ≥98-99%)
Impurity Management: Identifying and quantifying potential contaminants
Analytical Verification: Implementing comprehensive testing protocols
Documentation Requirements: Mandating detailed Certificates of Analysis (COAs)
These standards serve multiple critical functions in scientific research. Quality management systems ensure that peptides meet consistent performance metrics, enabling reproducible experimental outcomes. Researchers depend on these standardized protocols to maintain experimental validity and prevent potential data contamination.
Rigorous quality control is not optional – it is the foundation of credible scientific investigation.
Peptide suppliers must adhere to strict protocols that cover the entire product lifecycle, from initial synthesis through final quality verification. This involves implementing advanced analytical techniques like high-performance liquid chromatography (HPLC), mass spectrometry, and nuclear magnetic resonance (NMR) spectroscopy to confirm molecular structure and purity.
Pro tip: Always request and thoroughly review the Certificate of Analysis before purchasing research peptides to ensure they meet your specific experimental requirements.
Categories of Peptide Suppliers and Products
The research peptide market encompasses diverse supplier categories, each serving unique scientific requirements. Reliable peptide procurement demands understanding these distinct supplier segments and their specialized product offerings.
Peptide suppliers can be broadly categorized into several key types:
Academic/Research Institutions: Specialized suppliers focused on custom peptide synthesis for scientific research
Commercial Biotechnology Companies: Large-scale manufacturers producing standardized peptide products
Contract Manufacturing Organizations (CMOs): Providing peptide synthesis services for pharmaceutical and research clients
Specialized Peptide Research Vendors: Focused on niche or highly specific peptide formulations
Pharmaceutical Research Suppliers: Offering peptides for drug development and clinical research
These suppliers typically provide various peptide product categories, including:
Synthetic research peptides
Therapeutic peptide analogs
Custom-sequence peptides
Labeled and modified peptides
Reference standard peptides
Peptide libraries for screening
Not all peptide suppliers are created equal – research integrity starts with selecting the right source.
Product selection requires careful consideration of research peptide market dynamics, including purity levels, synthesis methods, and intended research applications. Researchers must evaluate suppliers based on their specific experimental needs, considering factors like molecular weight, amino acid composition, and potential modifications.
Pro tip: Prioritize suppliers offering comprehensive Certificates of Analysis and demonstrating transparent third-party testing protocols to ensure research-grade peptide quality.
Purity, Identity, and Quality Assurance Benchmarks
Research peptide quality hinges on rigorous purity, identity, and quality assurance benchmarks that define the fundamental standards for scientific investigation. Peptide reference standards provide critical frameworks for ensuring reproducible and reliable research outcomes.
Key quality assurance benchmarks typically encompass multiple critical dimensions:
Molecular Purity: Minimum 98-99% chemical composition
Structural Integrity: Confirmed amino acid sequence and configuration
Contaminant Screening: Comprehensive impurity detection protocols
Mass Spectrometry Verification: Precise molecular weight confirmation
Chromatographic Analysis: High-performance liquid chromatography validation
Analytical techniques for establishing peptide quality include:
Nuclear magnetic resonance (NMR) spectroscopy
Mass spectrometry profiling
High-performance liquid chromatography (HPLC)
Amino acid compositional analysis
Stability and degradation studies
Quality is not an afterthought – it is the foundation of credible scientific research.
Pharmaceutical quality control demands meticulous verification processes that go beyond basic chemical analysis. Researchers must evaluate peptides through comprehensive testing that confirms not just purity, but also long-term stability, potential interactions, and consistent performance across multiple experimental conditions.

Here’s a comparison of analytical methods used for peptide quality assurance:
Method | Purpose | Main Advantage |
HPLC | Purity and identity check | High sensitivity, quantitative |
Mass Spectrometry | Molecular weight confirmation | Detailed structure verification |
NMR Spectroscopy | Sequence and structure proof | Non-destructive, detailed data |
Amino Acid Analysis | Composition verification | Accurate content determination |
Pro tip: Request complete Certificates of Analysis that detail multiple verification metrics, not just a single purity percentage.
Good Manufacturing Practice (GMP) and ISO Standards
Research peptide production demands extraordinary precision, with Good Manufacturing Practice (GMP) and ISO standards serving as the critical regulatory frameworks ensuring product quality and scientific integrity. Peptide manufacturing standards establish comprehensive guidelines that govern every aspect of peptide production and research.
Key components of GMP and ISO standards include:
Facility Design: Controlled environmental conditions
Personnel Qualifications: Mandatory training and expertise requirements
Documentation Protocols: Rigorous record-keeping and traceability
Quality Management Systems: Systematic process controls
Continuous Monitoring: Regular performance and quality assessments
The specific dimensions of these standards encompass multiple critical areas:
Equipment calibration and maintenance
Staff competency and certification
Comprehensive risk management procedures
Analytical method validation
Impurity tracking and mitigation
Regulatory compliance is not a checkbox – it’s a commitment to scientific excellence.
Implementing these standards requires meticulous attention to detail, with organizations needing to develop robust quality management systems that integrate comprehensive testing, documentation, and continuous improvement processes. Researchers must understand that these frameworks are not merely bureaucratic requirements but fundamental mechanisms for ensuring reproducible and reliable scientific outcomes.
Pro tip: Request detailed documentation demonstrating a supplier’s GMP and ISO certifications before committing to peptide procurement.

Documentation, Traceability, and Compliance Expectations
Research peptide documentation represents the critical backbone of scientific credibility, establishing a comprehensive framework for transparent research practices. Systematic documentation ensures that every step of peptide production, from initial synthesis to final quality verification, can be meticulously tracked and independently validated.
Key documentation requirements encompass multiple essential components:
Chain of Custody: Complete tracking from raw material sourcing
Batch-Specific Records: Detailed manufacturing process documentation
Analytical Test Reports: Comprehensive verification data
Temperature and Storage Logs: Environmental condition tracking
Personnel Qualification Documents: Staff competency verification
Traceability standards demand rigorous documentation practices, including:
Unique batch identification codes
Time-stamped production records
Raw material origin certificates
Instrument calibration documentation
Detailed deviation and investigation reports
Documentation is not bureaucracy – it’s the scientific method in practice.
Compliance expectations require researchers and suppliers to maintain meticulous records that demonstrate unbroken evidence of product quality, handling, and integrity. These documentation protocols serve as the fundamental mechanism for ensuring reproducibility, accountability, and scientific trust.
Pro tip: Implement a digital documentation management system that allows real-time tracking, instant retrieval, and comprehensive audit trail capabilities.
Common Pitfalls When Selecting a Supplier
Research peptide procurement involves navigating complex quality landscapes, with supplier selection challenges presenting significant risks to scientific research integrity. Understanding these potential pitfalls is crucial for maintaining experimental reproducibility and data reliability.
Common red flags when evaluating peptide suppliers include:
Inconsistent Purity Documentation: Vague or missing Certificates of Analysis
Lack of Third-Party Verification: Absence of independent testing results
Insufficient Traceability: Unclear origin of raw materials
Minimal Quality Control Protocols: Limited analytical testing procedures
Inadequate Batch Consistency: Variable product performance across batches
Researchers should be particularly cautious of suppliers demonstrating these warning signs:
No detailed manufacturing process documentation
Limited or non-existent technical support
Pricing significantly below market standards
Inability to provide comprehensive molecular characterization
Lack of regulatory compliance certifications
Cutting corners on supplier selection is a direct threat to scientific credibility.
Pharmaceutical quality standards emphasize the critical importance of rigorous supplier evaluation. Researchers must conduct thorough due diligence, examining not just product specifications, but the entire quality management infrastructure of potential peptide suppliers.
This table summarizes potential risks and strategies when evaluating peptide suppliers:
Pitfall | Impact on Research | Mitigation Strategy |
Vague documentation | Unverifiable peptide quality | Request detailed Certificates of Analysis |
No third-party testing | Risk of hidden contaminants | Choose suppliers with independent verification |
Poor traceability | Unknown source, safety risks | Require origin and batch records |
Minimal quality protocols | Inconsistent product results | Review supplier quality assurance procedures |
Low pricing below market standards | Suspicion of inferior quality | Investigate quality controls and certifications |
Pro tip: Create a comprehensive supplier evaluation checklist that includes molecular characterization, traceability protocols, and independent testing requirements before finalizing any procurement decision.
Ensure Your Research Integrity with Premier Aminos High-Quality Peptides
Navigating complex industry standards for peptide suppliers can be challenging when research demands rigorous quality assurance, traceability, and documented purity. If you need peptides that meet or exceed 99 percent purity and come with comprehensive Certificates of Analysis, Premier Aminos is your trusted partner. We specialize in supplying research-grade peptides that align with GMP and ISO compliance to support your scientific integrity and reproducibility.
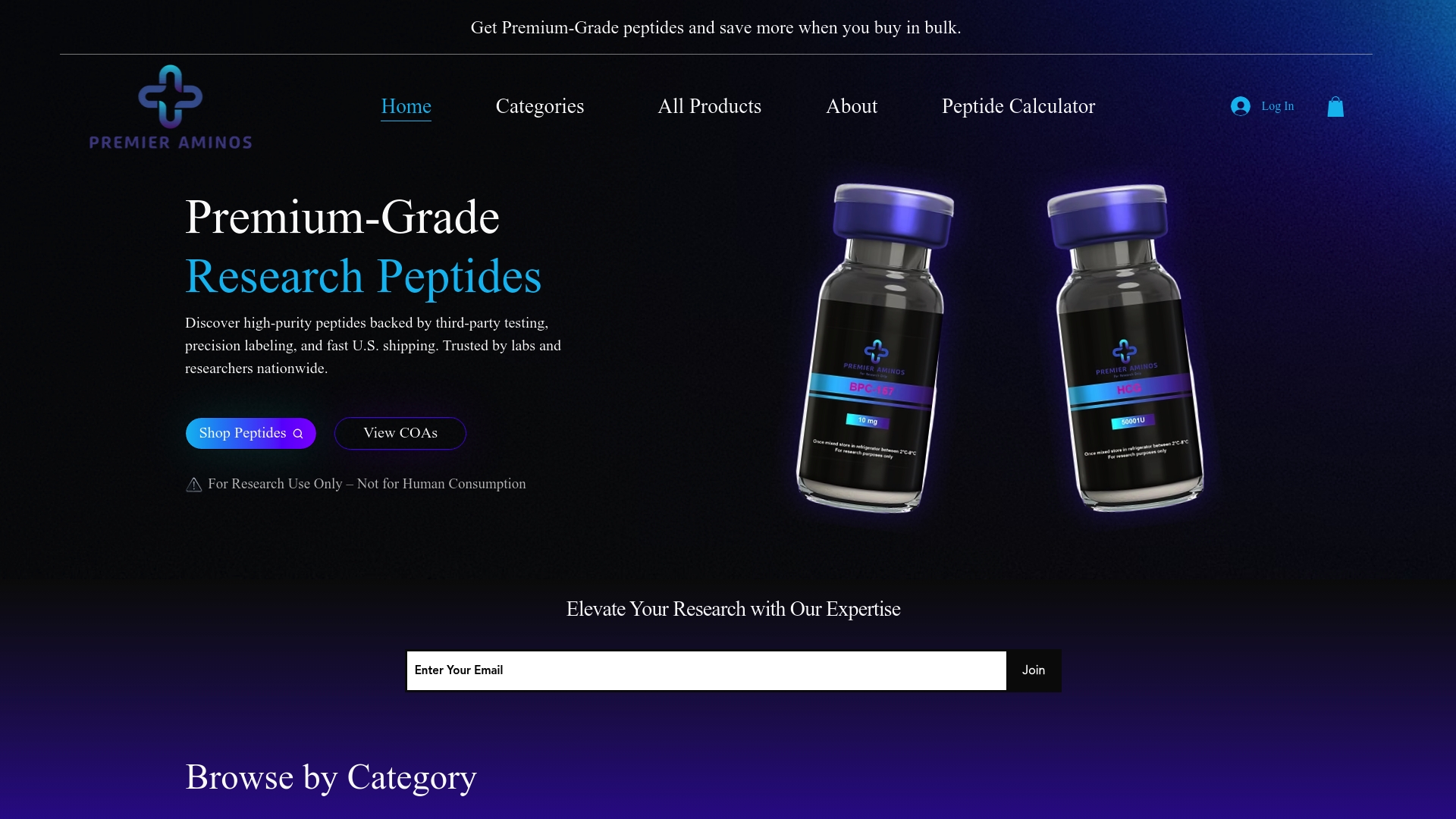
Explore our wide selection of expertly sourced Peptides | Premier Aminos including well-known compounds like GLP-1s and Tesamorelin designed for precision research needs. With fast U.S. shipping and transparent third-party lab testing, you can trust Premier Aminos to deliver reliable products that meet stringent documentation and quality benchmarks. Don’t compromise your experiments – visit Premier Aminos now to secure peptides that empower your research success.
Frequently Asked Questions
What are the key industry standards for peptide suppliers?
Industry standards for peptide suppliers include synthesis methodology, purity assessment (typically ≥98-99%), impurity management, analytical verification, and thorough documentation requirements, such as Certificates of Analysis.
How can I ensure the quality of research peptides before purchasing?
Request and review the Certificate of Analysis from suppliers, which should provide detailed purity metrics and analytical verification information, ensuring the peptides meet your specific experimental requirements.
What is the significance of Good Manufacturing Practice (GMP) in peptide production?
Good Manufacturing Practice (GMP) ensures that peptide products are consistently produced and controlled according to quality standards, emphasizing the importance of facility design, personnel qualifications, and rigorous documentation protocols to uphold scientific integrity.
Why is traceability important in the peptide supply chain?
Traceability is essential for maintaining product quality and scientific integrity, as it ensures that each peptide batch can be tracked from raw material sourcing through the entire manufacturing process, allowing for accountability and reproducibility in research.
Recommended
_edited.png)




Comments